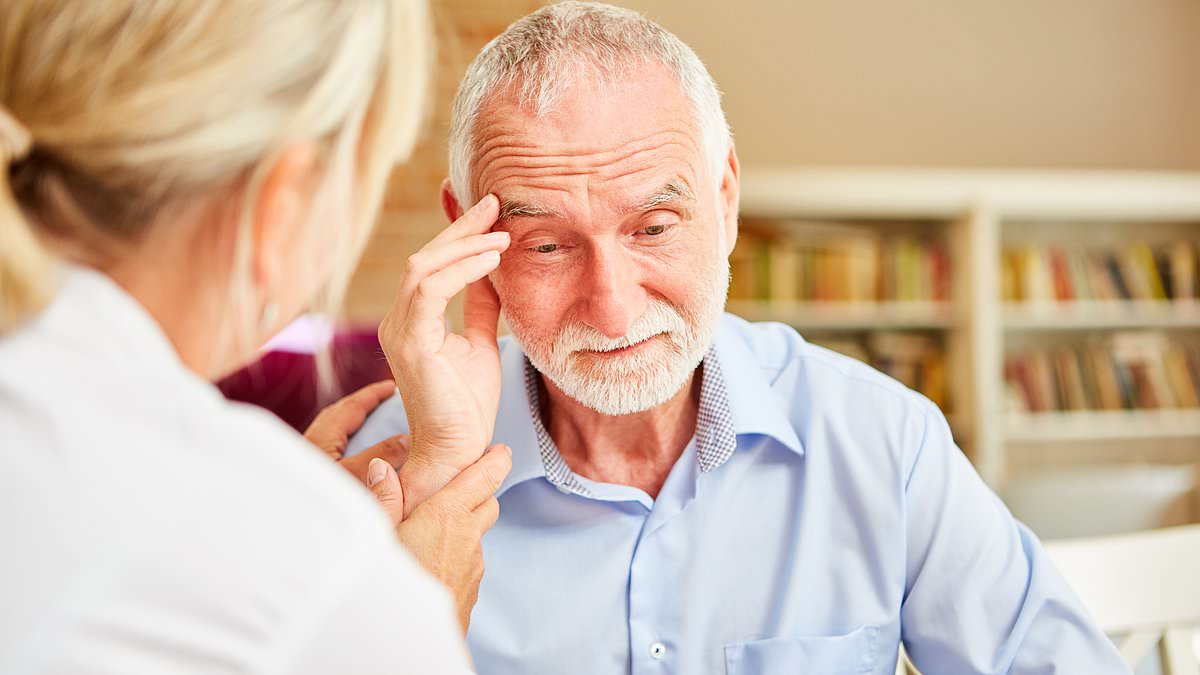
The US FDA has approved donanemab, a treatment for Alzheimer’s disease that is designed to slow the progression of early signs of the dementia.
Made by Indianapolis-based Eli Lilly, donanemab is a monoclonal antibody treatment that helps the body remove the hallmark plaque that builds up in the brain and causes dementia.
Clinical trials have shown the treatment slowed cognitive decline in patients by up to 35 percent.
Now approved, Eli Lilly said it will be sold under the name Kisunla and will be administered by once-monthly IV infusions. It will cost $695 per vial – amounting to about $32,000 a year.
While a breakthrough in Alzheimer’s care, experts have raised concerns about donanemab due to known risks of brain bleeds, which killed several trial patients.

Made by Indianapolis-based Eli Lilly, donanemab is a monoclonal antibody treatment designed to slow the progression of early signs of Alzheimer’s disease

Donanemab will be sold under the name Kisunla and will cost $695 per vial – amounting to about $32,000 a year
Dr Howard Fillit, co-Founder and chief science officer at the Alzheimer’s Drug Discovery Foundation (ADDF), said in a press release: ‘This approval marks another step forward in evolving the standard of care for people living with Alzheimer’s disease that will ultimately include an arsenal of novel treatments, providing much needed hope to the Alzheimer’s community.
‘Diagnosing and treating Alzheimer’s sooner than we do today has the potential to meaningfully slow disease progression, giving patients invaluable time to maintain their independence for longer.’
Eli Lilly said the drug slowed cognitive and functional decline in patients by up to 35 percent compared to placebo over 18 months.
It also reduced patients’ risk of progressing to advanced stages of Alzheimer’s by up to 39 percent.
The trial also found donanemab reduced amyloid plaques in the brains of participants by an average of 61 percent at six months, 80 percent at 12 months and 84 percent at 18 months compared to placebo.
While donanemab has been hailed as a potential therapy, one-quarter of patients in Eli Lilly’s trial suffered brain swelling and three people died from brain swelling or bleeding that was attributed to the drug.
Additionally, patients who received donanemab had a slightly higher mortality rate — two percent, compared with 1.7 percent who received placebo.
As the aging population of the US continues to grow, so will the rates of dementia. Currently, an estimated 6.7million Americans have Alzheimer’s disease – the most common cause of dementia – the vast majority of whom are aged over 65.
By 2050, this number is projected to rise to nearly 13 million.

The above graph shows the estimated projection of Alzheimer’s disease patients in the US through 2060
While the main cause of Alzheimer’s disease is still debated, scientists believe the damage is likely to be the result of abnormal build-up of proteins – amyloid and tau – in and around brain cells.
In Alzheimer’s patients, amyloid proteins are not effectively cleared from the body and eventually form plaques in the brain. Tau proteins detach from neurons to form tangles.
Both of these can lead to neuron death, which makes it difficult to deliver signals throughout the brain.
Donanemab is designed to target amyloids. Infused into the body via IV, the monoclonal antibodies travel to the brain.
Once inside the organ, donanemab binds to toxic build-ups of amyloid plaque, which prompts immune cells, known as microglia, to clear them.
Patients stop taking donanemab once the amyloid accumulation has been cleared from their brain.
The treatment is meant for people with early symptomatic Alzheimer’s disease or mild cognitive impairment.
Donanemab is not the first monoclonal antibody to be FDA approved. Lecanemab, sold as Leqembi, is already being used to treat Alzheimer’s patients.
Despite its risks, patients and families in the donanemab trial said the new dementia drug finally offers sufferers a sliver of ‘hope.’
Jim Sirois, now 68, from, was diagnosed with early-onset Alzheimer’s in 2020 after struggling to speak and becoming so forgetful he couldn’t remember where he had been the day before.
Mr Sirois was enrolled in the drug arm of the clinical trial, which his wife told DailyMail.com in August 2023 had effectively halted his condition over the past year.
Sue Sirois, 65, said: ‘I compare Jim’s decline to a friend I know who was diagnosed with Alzheimer’s at age 56. She died recently, and only lasted six years, whereas Jim is now over three years in and he is now still pretty much the same as what he was.’
Mrs Sirois added: ‘I would say those who have an opportunity to get this drug should go for it and get it because what else do you have? What other hope do you have? This drug is the only hope that people have right now – there is no other hope for the disease.’